List of important terms and explanation of what they mean.
Activation energy
Activation energy is a chemical term that describes how fast a reaction can proceed in relation to temperature and other factors. The less activation energy, the faster the reaction. Enzymes can often lower the activation energy of a reaction.
Allele
An allele of a gene is a specific version of the gene. Many organisms have two alleles of each gene.
Amino acid
An amino acid is a specific chemical group of substances. The 20 biological amino acids are combined in different combinations in the long chains that make up a protein. All amino acids contain an amine group (-NH3) and carboxy group (-COO-).
Antibiotic
An antibiotic is a substance that acts against specific microorganisms, either killing or inhibiting their growth.
Base pairing
Base pairing is the chemical attraction that causes two DNA strands to pair if T is opposite A and G is opposite C.
Cell factory
A cell factory is a cell that produces a valuable product, usually as a result of genetic engineering.
Codon
A codon is three consecutive nucleotides in a gene. Each of the 64 possible codons codes for an amino acid, which becomes part of the protein that the codon’s gene encodes. Codons are also called triplets.
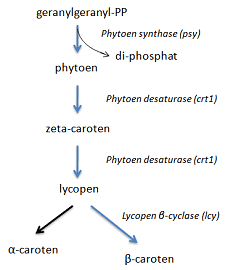
The genetic code
The genetic code is the translation of which amino acids the 64 possible codons lead to. See figure 1.3 in 1 What is DNA and genes?
DNA
DNA stores the genetic material (genes) of all cells in long double strands of nucleotides A, T, C and G. The order of these determines which protein the gene codes for.
DNA polymerase
A type of enzyme that can copy DNA in a process called replication.
Expression
In biology, expression is when a gene is activated and therefore leads to the creation of mRNA and then protein.
Expression vector
An expression vector is DNA that contains the auxiliary DNA elements (promoter, terminator etc) needed to express the gene in an organism. Read much more in 3 Genetic tuning.
Enzyme
Enzymes are proteins that catalyze certain biochemical reactions and processes, i.e. make them happen.
Fermentation
Fermentation is the cultivation of microorganisms in containers (fermenters). Read more.
Gel electrophoresis
Gel electrophoresis is a technique to separate different DNA or protein in a sample. Read more here.
Genome
A genome is an organism’s total amount of DNA sequence, i.e. all genes and gene regulatory regions etc.
Glycosylation
Proteins can be glycosylated by cells, which is an attachment of different kinds of sugars. Glycosylation can affect the half-life of a drug in the blood or, for example, provide specific signals about the protein to the cell.
Half-life
The half-life is the time it takes for the concentration of a substance to halve. Often used to refer to medication in the blood.
Heterozygous
A heterozygous organism has two different alleles of a gene, and the dominant form will be seen in the phenotype (how the organism turns out).
Homozygous
A homozygous organism has two identical alleles of the gene in question. If the alleles are the recessive (declining) form, they will, like the dominant form, be visible in the phenotype (how the organism turns out).
Catalysis
Catalyzing a reaction means increasing its ability to proceed (preferably significantly). The catalysis is done by a catalyst (e.g. an enzyme) that is not consumed in the reaction itself.
Complementary nucleotides
Nucleotides that fit together by base pairing. I.e. A-T and C-G.
Chromosome
Chromosomes are long strands of DNA. In normal (mitotic) cell division, one copy of each chromosome is transferred to the new cell.
mRNA
mRNA (messenger RNA) is a type of RNA. It is formed by transcription of the genes that are active and is used in translation to form protein.
Mutation
A mutation is a change in the DNA sequence that can occur to the advantage or disadvantage of the cell, e.g. due to UV light. Cells that are genetically altered are called mutants.
Nucleotide
The chemical structure that makes up the DNA and RNA strands. There are A, T, C and G. In RNA, T is replaced by the nucleotide U.
Protein
A protein is chemically a long chain of amino acids that are folded in a specific way to perform a function in the cell.
Recombinant production
Production with genetically modified organisms.
Replication
Replication is a cell biological process where a DNA strand is copied in two. A DNA double strand is separated along the way into two single strands, each of which has the complementary nucleotides attached. The result is two double strings.
Restriction enzyme
A restriction enzyme (or restriction endonuclease) is able to cut a DNA double strand at a specific sequence.
Screening
Screening is a method that can be used to distinguish specific genetic variants from the others.
Selection
Selection is a method of establishing conditions for organisms that allow them to survive and thus be selected. Read more.
Southern blot
Southern blotting is used to search for the presence of a specific DNA sequence in a DNA sample by base pairing with a probe. Read more.
Transgenic organism
An organism that contains genes from a foreign organism.
Transcription
Transcription is a process where active genes are transcribed from the DNA into mRNA, which can be used in the cell for translation.
Translation
Translation is a process where mRNA is translated into protein in a ribosome. Each codon (three nucleotides) is translated into an amino acid according to the genetic code.
Expressions
View expression